Protein Structure
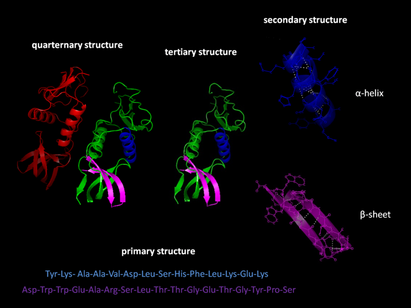
Proteins are not just limited to their "carbon backbone + molecules" structure system. In fact, proteins can actually take on four different levels of structuring, each one more complex than the last. These four levels are:
- Primary Structure
Primary structure is the least complex level of a protein. At this stage, the protein is merely a polypeptide chain of varying amino acids, in virtually any available order. These amino acids are attached via disulfide bonds. The type of protein varies on the order and type of amino acid. A protein composed of Serine, Leucine, and Alanine - in that order - is a very different protein from one composed of Lysine, Serine, and Asparagine. This linear binding and flexibility allows for the levels of complexity proteins may have.
- Secondary Structure
The second level of proteins. Secondary Structure in of itself has two forms - α-helices and ß-sheets. Both forms hold a common factor in production - hydrogen bonding between amino acids. α-helices are formed when the carbonyl oxygen molecules of the polypeptide backbone of the protein undergo hydrogen bonding with every fourth amino acid; this bonding pattern causes the helix shape of the protein. α-helices are components of most proteins in the body, along with ß-sheets. α-helices are also involved in the protein synthesis of DNA synthesis. ß-sheets are formed when the peptide strands of a polypeptide chain lie parallel to one another, and hydrogen bonding occurs between the carbonyl oxygen and the amine hydrogen. ß-sheets are also important components of most proteins in the body, and tends to be the most favored form of certain amino acids such as tryptophan, tyrosine and phenylalanine, and beta-branched amino acids like isoleucine, valine, and threonine.
- Tertiary Structure
The third level of proteins. Tertiary structure of proteins is achieved when the polypeptide chain of a primary of secondary structure protein folds and refolds upon itself, and the R-groups of its amino acids - the amine or amide groups, which determines the type of amino acid - undergo hydrogen bonding, ionic bonding, covalent bonding (disulfide bridges) or Van der Waals interactions. This causes the protein to exhibit 3-dimensional qualities, unlike the 2-dimensional secondary and primary proteins. Hydrophobic amino acids will face towards the core of the protein, to stay away from water. Tertiary proteins, on average, can contain as many as 300 to 400 amino acids. Tertiary proteins are components of many more complex proteins in the body - and Quarternary Proteins.
- Quarternary Structure
The fourth and final level of proteins, and the most complex. Quarternary structure of proteins is achieved in a much less complicated but much more complex method of formation - simply, two or more polypeptide chains, be it primary, secondary, or tertiary (but usually tertiary) undergo hydrogen, ionic, or other intramolecular bondings or interactions with each other, forming a complex 3-dimensional protein. Usually, two or more polypeptide chains will interact and each adopt a tertiary structure and then assemble with each other via intermolecular interactions. Quarternary proteins, due to this, are known as multimeric proteins; proteins with one polypeptide chain are known as monomeric proteins.Most proteins in the body are composed of quarternary structure, such as hormones (like HGH), muscles, bone, and other important molecules such as hemoglobin.
Proteins are not just limited to their "carbon backbone + molecules" structure system. In fact, proteins can actually take on four different levels of structuring, each one more complex than the last. These four levels are:
- Primary Structure
Primary structure is the least complex level of a protein. At this stage, the protein is merely a polypeptide chain of varying amino acids, in virtually any available order. These amino acids are attached via disulfide bonds. The type of protein varies on the order and type of amino acid. A protein composed of Serine, Leucine, and Alanine - in that order - is a very different protein from one composed of Lysine, Serine, and Asparagine. This linear binding and flexibility allows for the levels of complexity proteins may have.
- Secondary Structure
The second level of proteins. Secondary Structure in of itself has two forms - α-helices and ß-sheets. Both forms hold a common factor in production - hydrogen bonding between amino acids. α-helices are formed when the carbonyl oxygen molecules of the polypeptide backbone of the protein undergo hydrogen bonding with every fourth amino acid; this bonding pattern causes the helix shape of the protein. α-helices are components of most proteins in the body, along with ß-sheets. α-helices are also involved in the protein synthesis of DNA synthesis. ß-sheets are formed when the peptide strands of a polypeptide chain lie parallel to one another, and hydrogen bonding occurs between the carbonyl oxygen and the amine hydrogen. ß-sheets are also important components of most proteins in the body, and tends to be the most favored form of certain amino acids such as tryptophan, tyrosine and phenylalanine, and beta-branched amino acids like isoleucine, valine, and threonine.
- Tertiary Structure
The third level of proteins. Tertiary structure of proteins is achieved when the polypeptide chain of a primary of secondary structure protein folds and refolds upon itself, and the R-groups of its amino acids - the amine or amide groups, which determines the type of amino acid - undergo hydrogen bonding, ionic bonding, covalent bonding (disulfide bridges) or Van der Waals interactions. This causes the protein to exhibit 3-dimensional qualities, unlike the 2-dimensional secondary and primary proteins. Hydrophobic amino acids will face towards the core of the protein, to stay away from water. Tertiary proteins, on average, can contain as many as 300 to 400 amino acids. Tertiary proteins are components of many more complex proteins in the body - and Quarternary Proteins.
- Quarternary Structure
The fourth and final level of proteins, and the most complex. Quarternary structure of proteins is achieved in a much less complicated but much more complex method of formation - simply, two or more polypeptide chains, be it primary, secondary, or tertiary (but usually tertiary) undergo hydrogen, ionic, or other intramolecular bondings or interactions with each other, forming a complex 3-dimensional protein. Usually, two or more polypeptide chains will interact and each adopt a tertiary structure and then assemble with each other via intermolecular interactions. Quarternary proteins, due to this, are known as multimeric proteins; proteins with one polypeptide chain are known as monomeric proteins.Most proteins in the body are composed of quarternary structure, such as hormones (like HGH), muscles, bone, and other important molecules such as hemoglobin.
